Tag: bioequivalence
Discover why generic drugs cost 80% less than brand names. This guide explains bioequivalence, the Hatch-Waxman Act, and real 2025-2026 pricing data to help you save money safely.
Explore the complex challenges of proving bioequivalence for combination products, from FDC interactions to drug-device user interface hurdles and regulatory traps.
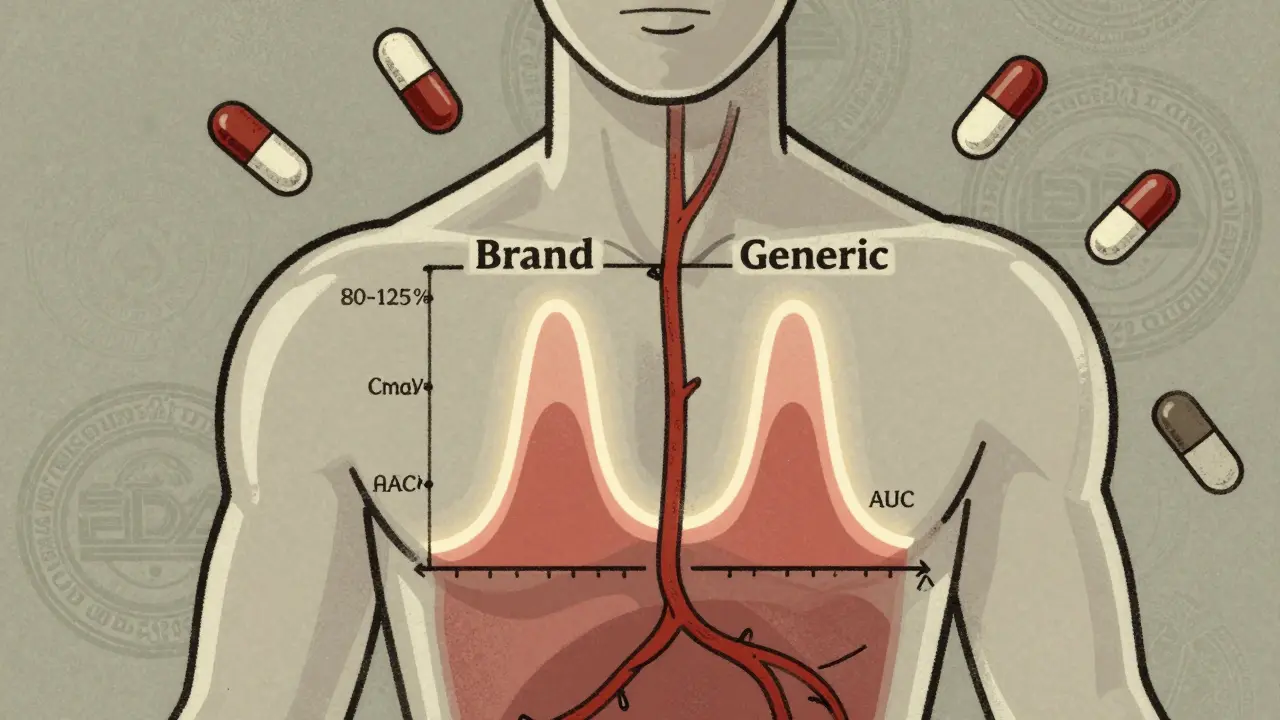
Learn how the FDA ensures generic drugs work just like brand-name versions through strict bioequivalence testing. Understand the science behind Cmax, AUC, and the 80-125% rule that guarantees safety and effectiveness.
An ANDA is the FDA's pathway for approving generic drugs without repeating costly clinical trials. It saves patients billions annually by ensuring safe, effective copies of brand-name medications enter the market quickly.
Cmax and AUC are the two key metrics used to prove generic drugs work the same as brand-name versions. Cmax measures peak concentration, while AUC measures total exposure. Both must fall within 80%-125% to ensure safety and effectiveness.